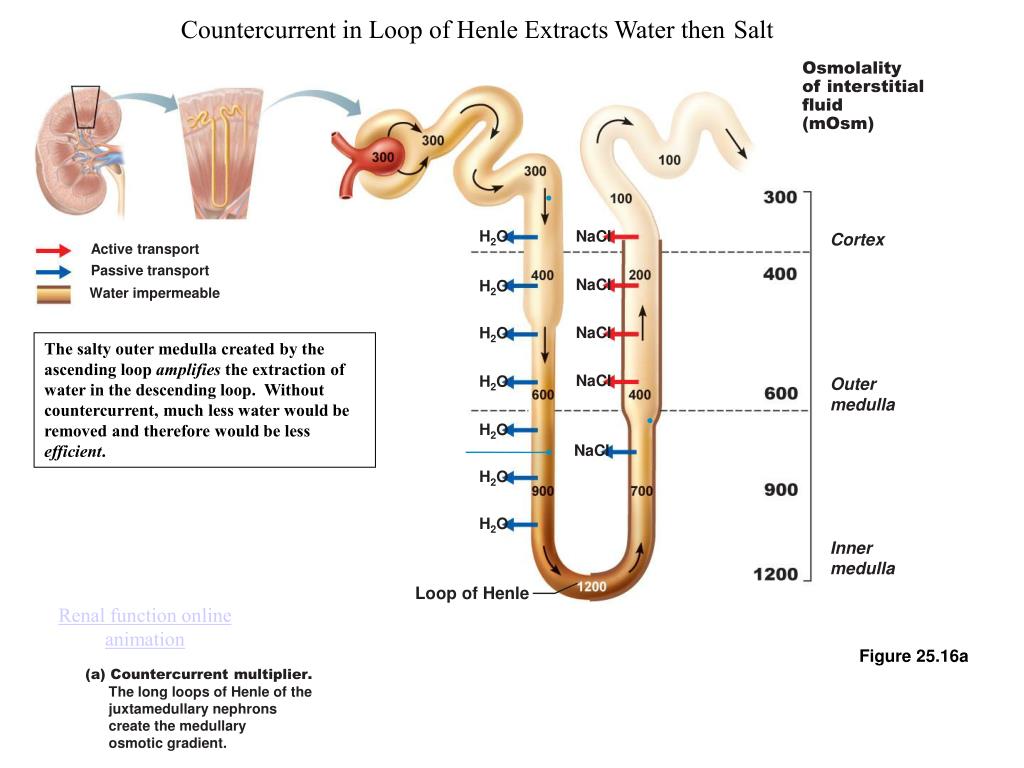
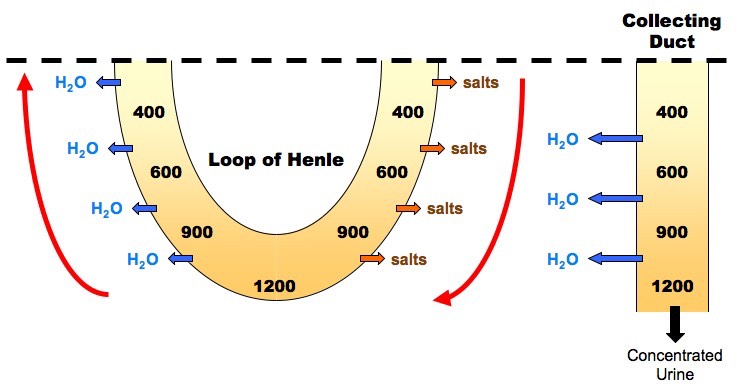
Two types of K + channels are found in the apical membranes of distal nephron cells.

(2012) reported that in more proximal parts of the distal nephron, identified as “DCT2/CNT” segments, ENaC activity is more constitutive and virtually independent of aldosterone. Indeed, in rats and mice under control conditions (normal rodent chow), ENaC activity is very low in the collecting ducts and CNTs, suggesting minimal K + secretory function of those segments ( Frindt and Palmer, 2004a Frindt et al., 2009 Frindt and Palmer, 2010). They are strongly regulated by the adrenal corticosteroid aldosterone, which is a major determinant of renal K + excretion ( Garty and Palmer, 1997 Verrey et al., 2008).
Na + channels in the kidney are identified as the epithelial Na channel (ENaC ). Apical Na + permeability plays a major role in determining the overall rate of K + secretion ( Weinstein, 2005b Frindt et al., 2009). Apical Na + channels mediate Na + entry into the cells from the lumen, stimulating K + entry across basolateral membranes by activating the Na +/K + ATPase and K + exit into the urine through depolarization of the apical membrane. K + enters the cells from the interstitial fluid mainly through the Na/K ATPase and exits the cells through apical K + channels. Widely accepted cellular models for secretion include three essential transport elements. Classical micropuncture experiments done in the 1960s indicated that most K + secretion takes place in the distal nephron, including the distal convoluted tubule (DCT) and the downstream connecting tubules (CNTs)-the “micropuncture accessible” portion ( Malnic et al., 1964). Proximal nephron segments, especially the proximal convoluted tubule and the ascending limb of Henle’s loop, reabsorb most of the K + filtered by glomeruli, leaving the distal nephron segments to secrete or reabsorb additional K + to match the rate of absorption of the ion from the gut ( Malnic et al., 2008). The kidneys excrete K + into the urine to maintain balance with dietary intake. Therefore, adaptation to increased K + intake involves the extension of robust K + secretion to more distal parts of the nephron. The enhanced secretion of K + by the CNTe and the recruitment of secretion by the CNTas account for the additional transport required for K balance. In animals fed a diet supplemented with 3% K, ENaC currents increased modestly in the CNTe but strongly in the CNTas, while ROMK currents tripled in both segments. Enhanced reabsorption by a separate mechanism is required to avoid excessive urinary K + losses. In animals fed a K-deficient diet (0.1% K), both ENaC and ROMK currents in the CNTe decreased by ∼50%, predicting a 50% decline in K + secretion. A mathematical model of the rat nephron suggested that K + secretion by the CNTe predicted from these currents provides much of the urinary K + required for K balance on this diet. ROMK activity, measured as TPNQ-sensitive currents, was substantial in both segments. This functional difference correlated with alterations in the intracellular location of ENaC, which was at or near the apical membrane in CNTe and more cytoplasmic in the CNTas. In more distal parts of the CNT (aldosterone-sensitive portion ), these currents were minimal. Under basal dietary conditions (0.5% K), ENaC activity, measured as amiloride-sensitive currents, was high in cells at the distal end of the distal convoluted tubule (DCT) and proximal end of the connecting tubule (CNT), a region we call the early CNT (CNTe). We measured the activities of epithelial Na channels (ENaC) and ROMK channels in the distal nephron of the mouse kidney and assessed their role in the process of K + secretion under different physiological conditions.
